Resources
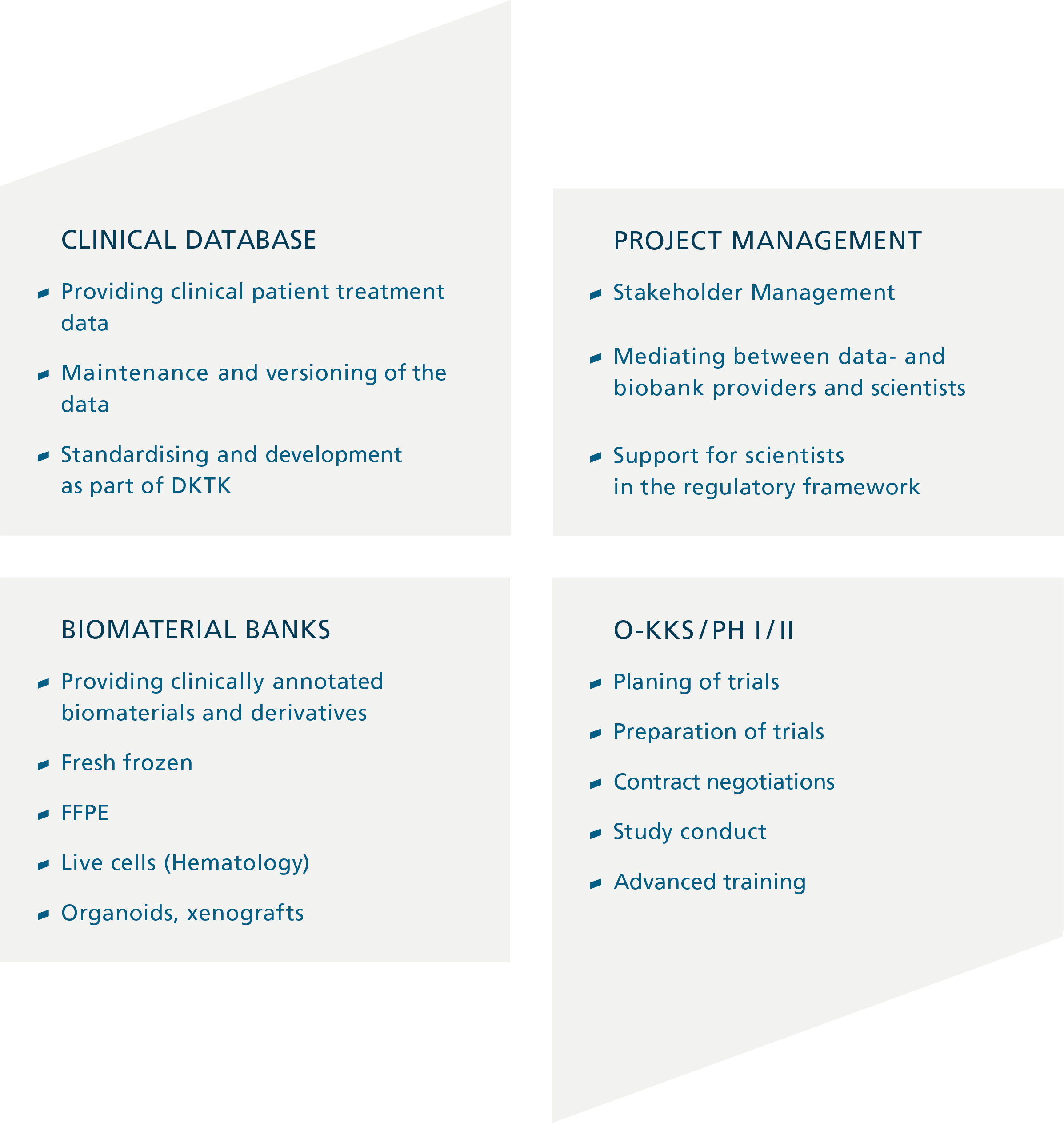
At UCT Frankfurt, a complete clinical data collection exists for all patients first diagnosed and treated at UCT since 2012 (approximately 10,000 patients). Biomaterials are available from patients whose tumor material was examined in pathology. Fresh frozen material is also available from over 8,000 patients. Moreover, a bone marrow biobank is connected, in which vital frozen bone marrow from leukemia patients is stored.
Since 2016, the establishment of an organoid biobank of patients with colorectal carcinoma and further entities has been started at the Georg-Speyer-Haus. A central access system and a project management team enable the efficient use of these resources by scientists via the Clinical communication platform (CCP) of the German Consortium for Translational Cancer Research (DKTK) The CCP, which is dedicated to the curation, expansion and scientific sharing of the clinically annotated biomaterial banks of the members of the DKTK and most of the German centers of excellence in oncology, is coordinated from Frankfurt. The backbone of the CCP is an IT structure (bridgehead) that allows to search for patient cohorts across sites and to obtain data and biomaterials for scientific purposes while respecting the digital data rights of patients and treatment centers. The cohort is growing rapidly, and currently includes data on approximately 650,000 cancer patients. For the majority of these patients, biomaterials are available in the affiliated local biomaterial banks and pathology archives.
In addition, the FCI has a central unit, the Oncology Clinical Trials Coordination Center (O-KKS) at UCT, which supports all aspects of self-initiated studies. This unit was expanded in 2021 to become the Central Study Coordination (ZSK) of the Department of Medicine, and now coordinates, monitors and audits all self-initiated studies at Frankfurt University Medical Center across all indications on behalf of the department. The principle of subsidiarity applies here, which leaves the freedom of content and scientific design with the initiators, provides organizational and regulatory assistance where it is needed, and ensures the necessary, legally prescribed quality and safety management, which according to drug law is a task of the study sponsor, i.e. in the case of self-initiated studies, the department, represented by the department chair. Extensive experience exists in the coordination of academic studies of all phases; in a phase I/II study unit, specialized personnel perform complex study therapies including first-in-man studies.